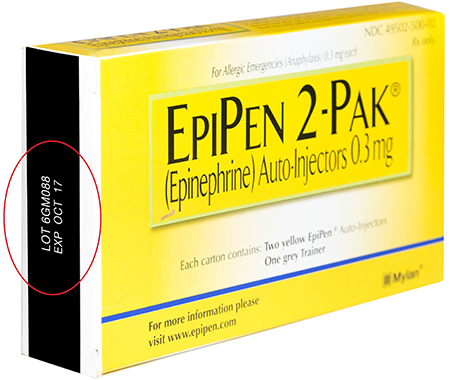
 Meridian Medical Technologies is recalling of 13 lots of Mylan’s EpiPen and EpiPen Jr Auto-Injector products used for emergency treatment of severe allergic reactions.
Meridian Medical Technologies is recalling of 13 lots of Mylan’s EpiPen and EpiPen Jr Auto-Injector products used for emergency treatment of severe allergic reactions.
The recalled devices may contain a defective part that could cause it to fail to activate. The recalled product was manufactured by Meridian Medical Technologies and distributed by Mylan Specialty.
Although the number of reported failures is small, the EpiPen products are being recalled because of the potential for life-threatening risk if a severe allergic reaction goes untreated, the U.S. Food and Drug Administration said.
Consumers should keep and use their current EpiPens until they get a replacement, the agency advises. Consumers should contact Stericycle at 877-650-3494.
As stated on the product label, consumers should always seek emergency medical help right away after using their EpiPens, particularly if the device didn’t activate.
At this time, the 13 lots identified – distributed between Dec. 17, 2015, and July 1, 2016 – are the only EpiPen lots included in the U.S. recall.
Consumers who have EpiPens from lots that aren’t included in this recall don’t need to replace their EpiPen before to the expiration date.
|
Product/Dosage |
NDC Number |
Lot Number |
Expiration Date |
|
EpiPen Jr Auto-Injector, 0.15 mg |
49502-501-02 |
5GN767 |
April 2017 |
|
EpiPen Jr Auto-Injector, 0.15 mg |
49502-501-02 |
5GN773 |
April 2017 |
|
EpiPen Auto-Injector, 0.3 mg |
49502-500-02 |
5GM631 |
April 2017 |
|
EpiPen Auto-Injector, 0.3 mg |
49502-500-02 |
5GM640 |
May 2017 |
|
EpiPen Jr Auto-Injector, 0.15 mg |
49502-501-02 |
6GN215 |
September 2017 |
|
EpiPen Auto-Injector, 0.3 mg |
49502-500-02 |
6GM082 |
September 2017 |
|
EpiPen Auto-Injector, 0.3 mg |
49502-500-02 |
6GM072 |
September 2017 |
|
EpiPen Auto-Injector, 0.3 mg |
49502-500-02 |
6GM081 |
September 2017 |
|
EpiPen Auto-Injector, 0.3 mg |
49502-500-02 |
6GM088 |
October 2017 |
|
EpiPen Auto-Injector, 0.3 mg |
49502-500-02 |
6GM199 |
October 2017 |
|
EpiPen Auto-Injector, 0.3 mg |
49502-500-02 |
6GM091 |
October 2017 |
|
EpiPen Auto-Injector, 0.3 mg |
49502-500-02 |
6GM198 |
October 2017 |
|
EpiPen Auto-Injector, 0.3 mg |
49502-500-02 |
6GM087 |
October 2017 |
Health care professionals and consumers should report any adverse reactions or device malfunctions to the FDA’s MedWatch program by one of the following:
- Completing and submitting the report online at www.fda.gov/medwatch/report.htm.
- Downloading and completing the form, then submitting it by fax at 800-FDA-0178.





